 
The valency of elements from the same groups is the same. There is no change in valency among members of a group. Moving left to right across a period in the periodic table, valency increases first, then declines. Hydrogen, lithium, and sodium elements, for example, are all found in the first group and all have the same number of valence electrons, which is one.Īn atom’s valency is its ability to combine. It denotes that elements in the same group have the same number of valence electrons. Valence electrons remain consistent across a group. The amount of valence electrons grows as we move left to right across a period in the periodic table. The electrons in an atom’s outermost shell are referred to as the atom’s valence electrons. The explanation for this unusual behaviour is that in the case of noble gases, atomic radius relates to van der Waal’s radius, whereas in the case of other elements, it refers to the covalent radius. When compared to the preceding halogen atom, the atomic radii of inter gases rapidly increase. Moving from top to bottom in a group, atomic radii gradually rise as nuclear charge and the number of shells grows.Įxception: Noble gases exhibit extraordinary behaviour. This is due to the fact that as we move left to right in a period, the atomic number of the elements increases, causing the nuclear charge to increase while the number of shells in elements remains constant. 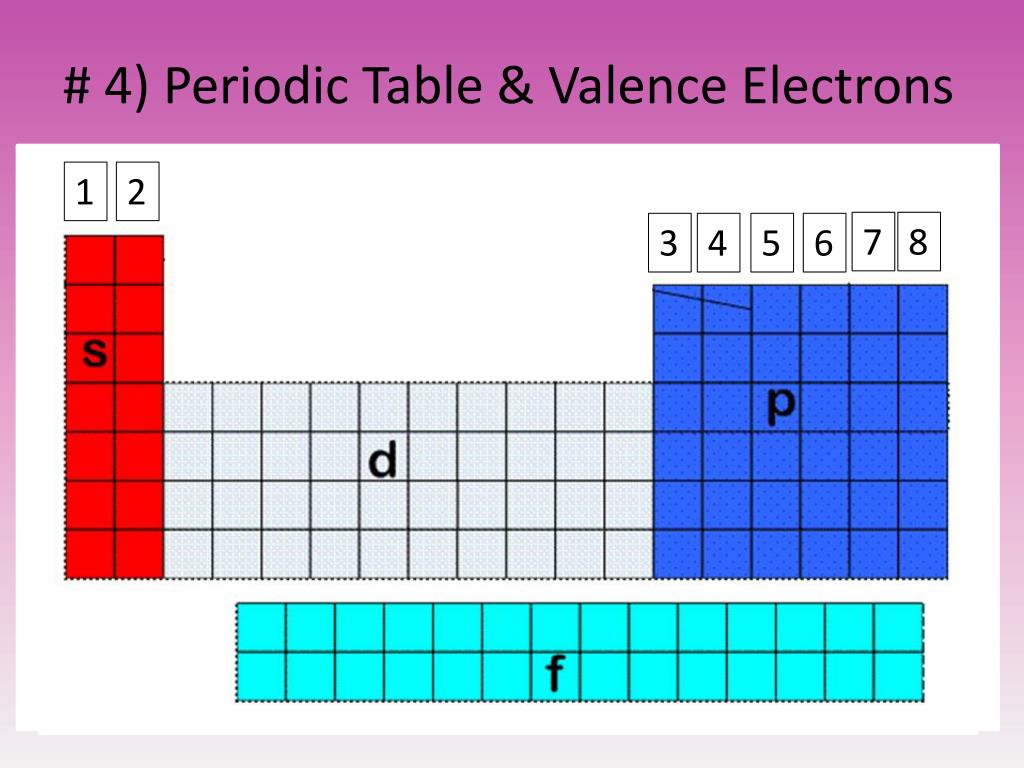
The periodic trend of atomic radius across a period – Atomic radius steadily lowers as we move from left to right in a period. The atomic radius is the distance between an atom’s nucleus and its outermost shell. In modern-day, scientists using Mendeleev’s Periodic Table as a base created a long-form periodic table which is called Modern Periodic Table. But in 1863 Russian chemist Dmitri Mendeleev, gave Mendeleev’s Periodic Table which is the most successful attempt of that time. There are various failed attempts to arrange the known elements so that all the elements with similar properties are arranged together. What is Periodic Table?Ī periodic Table is the arrangement of all the elements in the known universe based on either atomic weight or atomic number. So, let’s learn about these trends in the properties of elements while arranged in the periodic table. This article will explore the same thing with the modern-day lens and will tell you about all the trends in various properties such as atomic radius, valency, metallic or non-metallic characters, reactivity, boiling point, melting point etc. Scientists in the early days observed that while arranging the elements based on either atomic weight or atomic number, elements with similar properties automatically got arranged closer to each other. ISRO CS Syllabus for Scientist/Engineer ExamĪrticle with the name “Periodic Table Trends” as the name suggests explores the trends and patterns in the property of elements while arranged in the modern-day periodic table.ISRO CS Original Papers and Official Keys. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
